 
You can collect carbon dioxide in the balloon or funnel. After mixing, you will observe the bubbles in the glass tube, and these bubbles are of carbon dioxide. Firstly, take one teaspoon of baking soda in the glass tube and put ¼ cup vinegar in it. The equation of this reaction is written below.Ĭalcium carbonate dilute hydrochloric acid → calcium chloride carbon dioxide water At HomeĬarbon dioxide can easily be prepared at home by baking soda and Vinegar. Due to the lightweight nature of hydrogen, it goes into the atmosphere fastly, and carbon dioxide gets collected in the funnel. When we mix the calcium carbonate with the dilute hydrochloric acid, the calcium chloride forms along with this, the carbon dioxide and hydrogen gas are also released. So, for industrial preparation methods, the calcium carbonate reacts with hydrochloric acid. Industrially, carbon dioxide is produced by reacting the carbonates with acids. These are the several natural methods of preparation of carbon dioxide. Carbon dioxide is also produced and collected for future use during this process.Ĭarbon dioxide is also released in the process of decay. In this process, organic sugar-containing food like grapes is stored for many years for preparing alcohol, wines, and beers. It is extracted from these minerals and stored.Īlong with this, naturally, the carbon dioxide is also prepared by the process of fermentation. 
These caves and mines are found as mineral-like magnesite, dolomite, and limestone. Natural MethodĬarbon dioxide is found in caves and mines. There are different methods of preparation of carbon dioxide. Along with this, it has various methods of preparation and uses of Carbon Dioxide. Then, further, it is absorbed by the plants and utilised by them for synthesising the food through the process of photosynthesis. Its solubility at the temperature of 25 ☌ is 1.45 g/L.Ĭarbon dioxide is exhaled by human beings in the process of respiration. The heat capacity of carbon dioxide is 37.135 J/K The structure of carbon dioxide is Trigonal. About Carbon dioxideĬarbon dioxide is the colourless acidic gas, having a molar mass of 44.009 g Also, one can prepare carbon dioxide at home, but in a smaller quantity only. 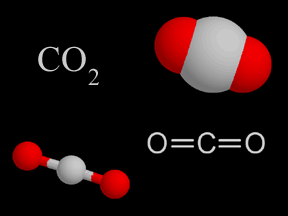
It is extracted from nature and can be prepared in laboratories. Along with the atmosphere, it is extracted manually for industrial and laboratory uses. 0.04% of the total gas in the atmosphere contains carbon dioxide. Its density is approximately 53% higher than the other gases present in the atmosphere. It is a type of chemical compound that originated in the form of colourless acidic gas. Carbon dioxide is widely used in industries, factories, and laboratories for performing various experiments. 08206 L atm mol -1 K -1, and T is the temperature measured in kelvin (K).The scientific formula of carbon dioxide is CO2. To account for these conditions, we use the ideal gas equation PV=nRT where P is the pressure measured in atmosphere(atm), V is the volume measured in liters (L), n is the number of moles, R is the gas constant with a value of. The reason why gases require additional information is because gases behave as ideal gases and ideal gases behave differently under different conditions. The mass we can find using stoichiometric conversions we have learned before. So we need either the temperature and volume, temperature and pressure, or pressure and volume. In order to solve for the temperature, pressure, or volume of a gas using chemical reactions, we often need to have information on two out of three of these variables. This is unlike regular solids where we only had to account for the mass of the solids and solve for the mass of the product by stoichiometry. For example, if we have a certain temperature or pressure, this can change the number of moles produced or the volume. 
However, a different approach is needed to solve reversible reactions.įor further clarification, when solving equations with gases, we must remember that gases behave differently under different conditions. \]īy using the Ideal Gas Law for unit conversions, properties such as the pressure, volume, moles, and temperature of a gas involved in a reaction can be determined. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
